How it works
GYPSOIL alters soil chemistry. Soil particles and organic matter have negative charges on their surfaces. Mineral cations (with positively charged surfaces) are attracted to these negative charges on the soil particles. The number of exchangeable cations that a soil is capable of holding and available for exchange with the soil water solution is called the CEC. This is an indication of the level of nutrients the soil can hold. Clay soils tend to have a high CEC.
GYPSOIL is calcium sulfate dihydrate (CaSO4 • 2H2O). It dissolves (dissociates) with moisture into free calcium and sulfate. The sulfate (SO4) attaches primarily to excess magnesium (Mg), aluminum (Al) and sodium (Na) in the soil complex, making soluble compounds that move down and out of the crop rooting environment. The remaining Ca from the gypsum then attaches to the exchange sites, replacing the excess Mg, Al, Na, etc. in the soil complex. This is what sets the stage for improved soil structure.
Influence of calcium
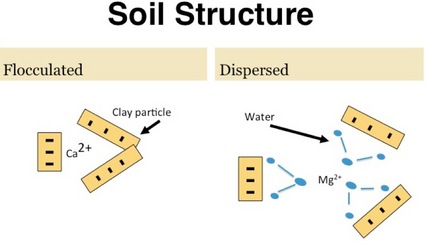
Calcium is a positively charged ion called a cation. Cations are absorbed by the plant roots and also held on exchange sites in soils. The positive charges of calcium are attracted to negative electrical charges found on the exchange sites on clay particles and Organic Matter (OM). The more clay and OM, the larger the attraction. This attraction of the positive Ca with negative charges in the clay particles -- binds the clay soil particles together so they become flocculated and resist dispersing and soil structure breakdown.
CEC
Cation Exchange Capacity or (CEC) is an indication of the volume of nutrients that can be held by the soil. One easy way to think of CEC is how big of a container the soil is for holding nutrients. The more organic matter and clay in the soil, the greater the CEC and the more nutrients can be held in it. Sandier soil typically has a lower nutrient-holding capacity and lower CEC. Trying to put ten gallons of nutrients into a five-gallon storage means nutrients will go to waste.
Cations are elements with one or more positive charges. Clay and organic matter are covered in negative electrical charges called exchange sites. Cations are attracted to and hold onto negative exchange sites. The amounts of different cations vary between soils. Generally, higher CEC soils require higher rates of GYPSOIL for soil amendment benefits. View our usage recommendations for GYPSOIL.
Reference: Hydrologic properties and leachate nutrient responses of soil columns collected from gypsum-treated fields Rebecca Tirado-Corbala´ *, Brian K. Slater, Warren A. Dick, Jerry Bigham, Edward McCoy.